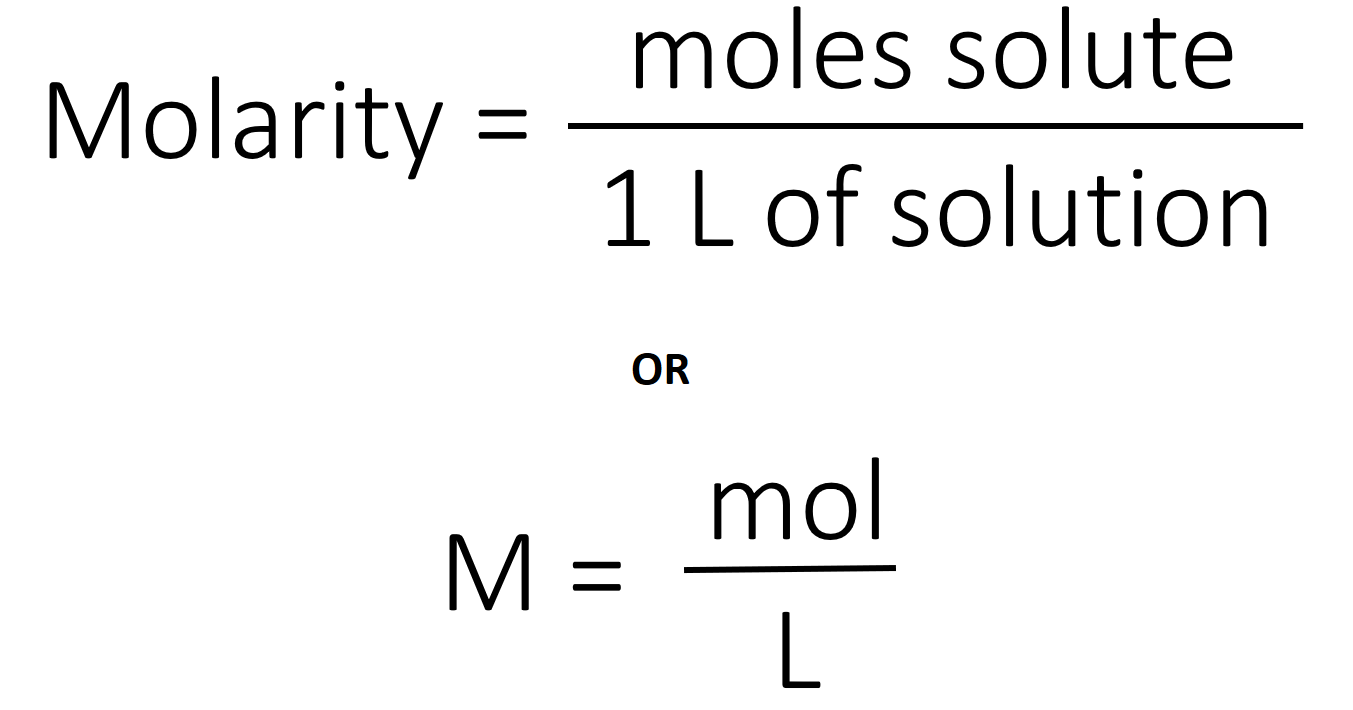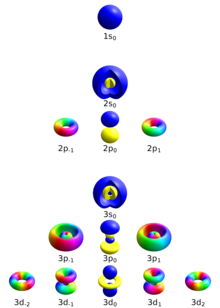What is the maximum number of orbitals that can be identified with the following quantum numbers ? n = 3, l = 1, ml = 0.

A pendulum of length ℓ = 1 m is released from theta 0 = 60 ^ ∘ . The rate of change of speed of the bob at theta = 30 ^ ∘is ( g = 10 m / s ^ 2 )

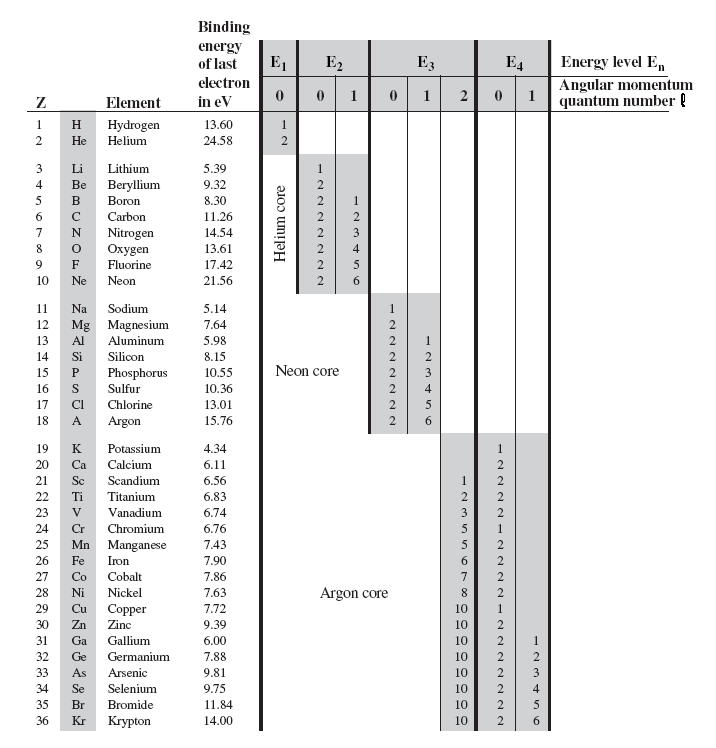
7.85 The electron configuration of a neutral atom is 1s22s22p63s2. Write a complete set of quantum numbers for each of the electrons. Name the element. - ppt download

What is the maximum number of orbitals that can be identified with the following quantum numbers ? n = 3, l = 1, ml = 0.

A pendulum of length ℓ = 1 m is released from theta 0 = 60 ^ ∘ . The rate of change of speed of the bob at theta = 30 ^ ∘is ( g = 10 m / s ^ 2 )
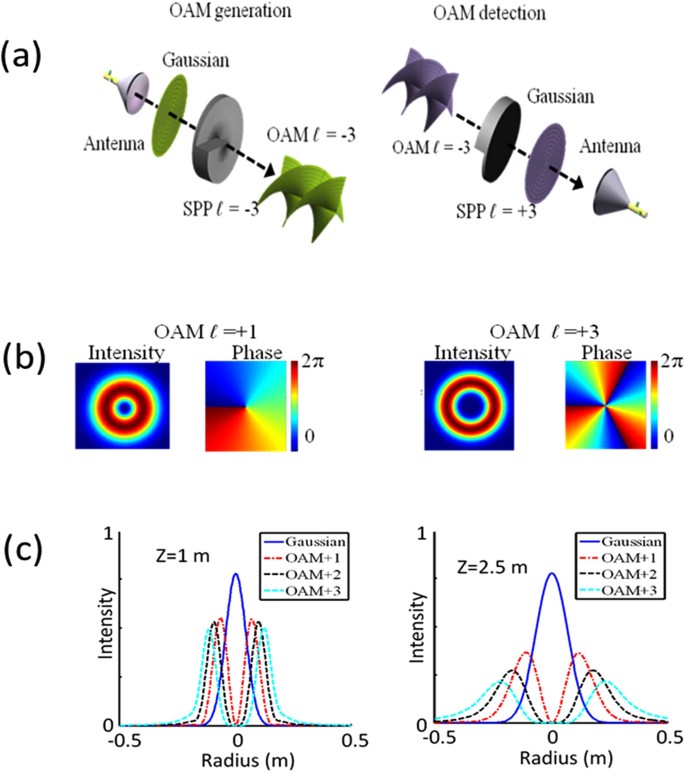
Multipath Effects in Millimetre-Wave Wireless Communication using Orbital Angular Momentum Multiplexing | Scientific Reports

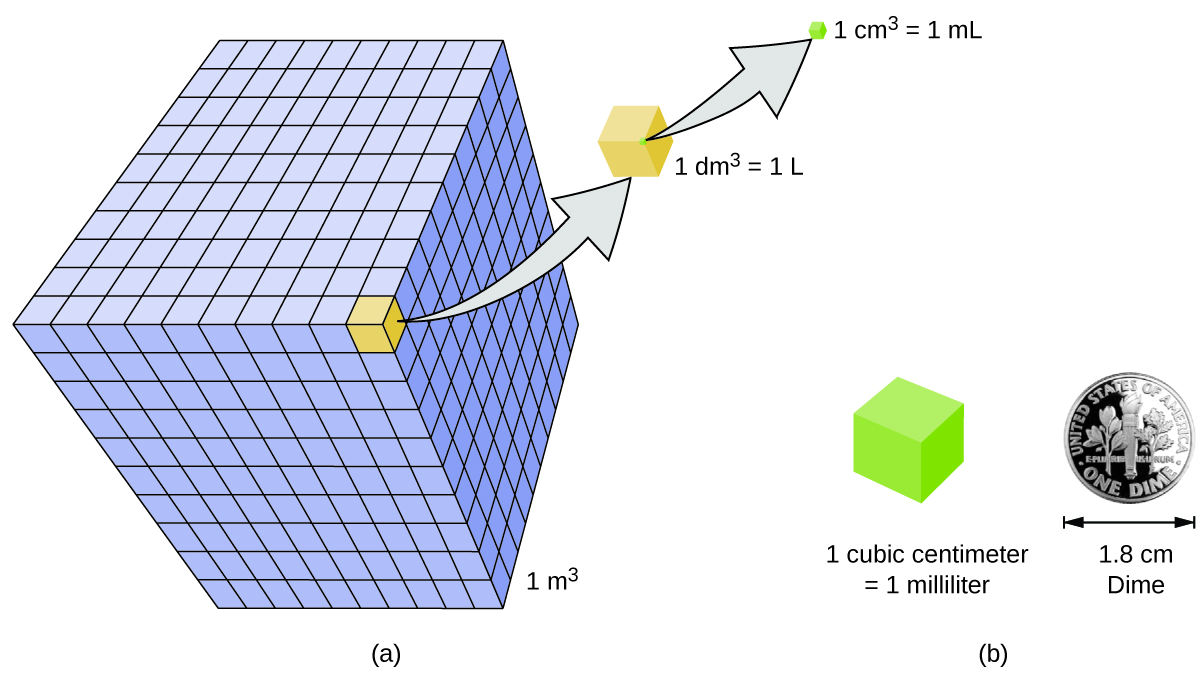
ACID-BASE CHEMISTRY. CONCENTRATION UNITS - I Mass concentrations Water analyses are most commonly expressed in terms of the mass contained in a liter. - ppt download